P Sunthar
-
sunthar@che.iitb.ac.in

Education
Courses
Research
Blood flow in coronary arteries
Constrictions in coronary arteries leads to lower supply of blood to the heart (ischemia) and is one of the leading causes of heart attacks. In order to predict the extent of ischemia it is important to model the artery network, the terminal boundary conditions. We use a combination of deterministic models of constrictions, combined with machine learning tools to predict the pressure drop across a series of constrictions. This can be used as a quick predictor of extent of constriction to help cardiologists to plan appropriate interventions.
Figure shows a depiction of major coronary arteries, stenosis locations and distribution of normalised pressure.

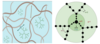
Colloidal fingering
Colloidal suspensions show spontaneous clustering instability. This can be understood using double-diffusive instability. Colloidal suspensions spontaneously form fingers on contacting a miscible solvent. . Fingers appear in shape to be similar to those seen in a thermohaline system. The initial interface must have a globally stable density stratification. Two solutes of varying diffusivities must have opposing effect on the mixture density.
Figure shows a graphic summary of colloidal finger formation under two common conditions leading to downward (left) and upward (right) moving fingers. The gray scale intensity indicates local mass density---dark being denser than less dense. Fingers move downward when the density of the bulk medium is in between that of the medium containing the colloid and the colloidal particle density. Fingers are expected to move upward when the colloidal particle density is lower than that of the bulk medium.

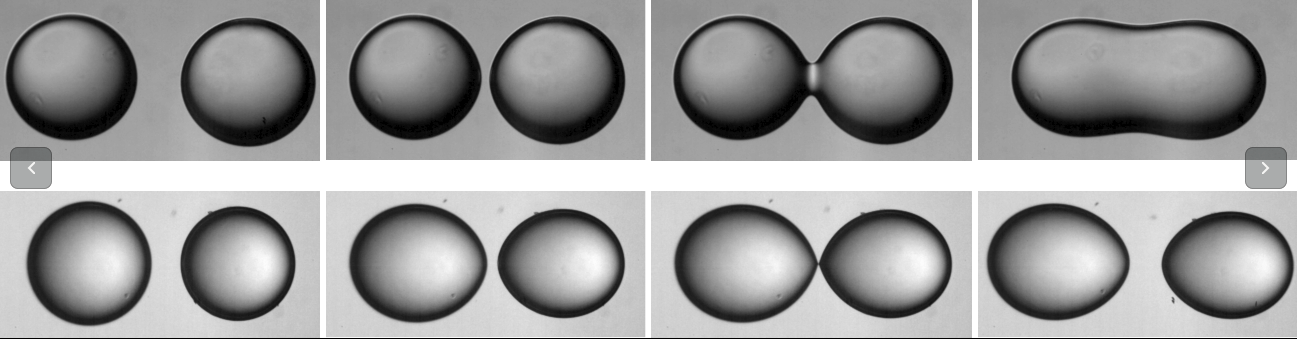
Self-assembly of vesicles in SPI method
Laboratory methods to produce unilamellar vesicles of a definite size either rely on an external energy input (such as sonication or extrusion), using multi-component systems (such as catanionic, lecithin+bile, etc.) or by inducing a spontaneous curvature through certain molecules. We have developed a method where lipids spontaneous assemble from a solution phase that results in predictable diameter vesicles. In this method stationary phases (one containing the lipid and other being aqueous) is brought into contact without any convective (advective) mixing. The time of synthesis of liposomes by this method can be made as low as 15 minutes. An equilibrium between a dilute and a condensed phase is easily observed under stationary conditions for single tailed surfactants, and diffusive mixing permits such an equilibrium, hitherto unobserved, in bilayer forming surfactants. While providing the quickest way to synthesize uniform unilamellar liposomes, this method may also help in identifying suitable component candidates and conjectures for the origin of cells in a prebiotic chemical environment that led to the origin of cellular compartments.

Diffusiophoretic enhancement of mass transfer
It is observed that nanoparticles enhance mass transfer of solute particles. While there have been many beliefs around what causes this apparent increase, there is no fundamental theory or experimental evidence to back any of them. We have reproduced the enhancement phenomenon within a glass capillary containing a dye solution on one side and nanoparticle suspension on the other, avoiding the inertial interference present in earlier experiments. The enhancement is explained by the convective motion (and not a diffusion) of the dye solution counter to the direction of the diffusiophoretic motion of the nanoparticles towards a higher concentration of the dye. The initial velocity of the dye front in the capillary and the dye-drop experiment agree quantitatively with independent theoretical estimates from the diffusiophoretic velocity of the nanoparticles.

Mucosal surfaces are the entry points of pathogens into the animal body and act as the primary sites of defense. Experiments show that viruses called bacteriophages undergo subdiffusive motion within mucus due to their adherence to the mucus network. However, the details of this interaction are unknown. This research aims to explain the motion of phages within mucus and its interaction with mucus networks using Brownian dynamics simulations of dendrimers in a network of associative polymers.
Rheology of wormlike micelles
Unentangled wormlike micellar solutions (WLMs) exhibit remarkable flow dynamics ranging from shear-thickening, shear-thinning and non-zero normal stress differences. The self-assembling of micellar solutions has practical applications in enhanced oil recovery and in drag reduction. However, the origins of their microstructural transitions and the rheological properties of these solutions are still poorly understood. We study model wormlike chain system that can spontaneously associate or dissociate to form varying length long polymeric chains. Hydrodynamic interactions (HI), which have been neglected in previous models seem to play an important part in explaining some behaviour in dilute and semi-dilute regimes.

Mechanical Properties of Biopolymer Networks
Mechanical forces play a crucial role in a cell's physiology and locomotion. Previous studies on cell mechanics have shown that the cell behaves as a viscoelastic, glassy, and inelastic material. However, a detailed understanding of the intermolecular origins of the mechanics and dynamics of the cells is still unknown. The cytoskeleton is a dynamic and active cross-linked biopolymer network, which governs the mechanical properties of cells. This research aims to understand cell mechanics through understanding the dynamics of the cytoskeletal biopolymer network. The biopolymer network will be modelled as a semidilute solution of sticky semi-flexible polymer chains, and Brownian dynamics are used to simulate the dynamics of the chains.
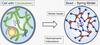
Brownian Dynamics Simulation
Our group is actively working with new algorithms to simulate polymer molecules of various architectures with hydrodynamic interactions. We are currently working on HOOMD Blue and Positively Split Ewald (PSE) for a nearly linear scaling of large polymer dynamics simulations.
We have an open Institute Post-Doctoral Fellowship (PDF) Position in the area of method development for HOOMD and PSE with GPUs (Nvidia's CUDA and AMD's HIP). Research scholars in the domain of computational physics are welcome to apply. This will also involve a visit to Monash Univiersity for a month in collaborative work with Prof. Ravi Jagadeeshan. Candidates with background in computational physics, molecular simulations and coding in C++, Python, GPU are encouraged to apply here:
Research Areas
Sub Areas
- Drug Delivery
- Polymer Physics
- Computational Flow Modelling (CFD)
- Fluid Mechanics and Stability
- Colloids
- Surfactants