Sarika Mehra
Core Faculty
Professor

112, Chemical Engineering
Core Faculty
Professor
Doctoral Dissertation Fellowship awarded to outstanding final-year PhD candidates University of Minnesota. (2004).
Best Thesis Award Indian Institute of Technology Delhi India. (1999)
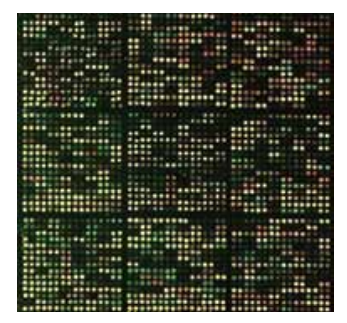
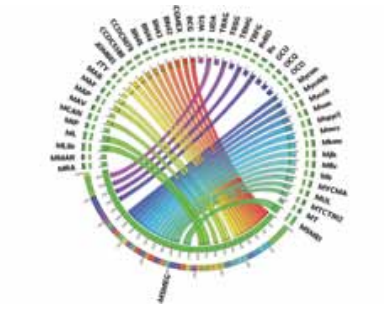
To identify mechanisms of resistance, our first model system is a nonpathogenic soil bacteria, Streptomyces coelicolor. Streptomyces species account for more than two-thirds of all antibiotics. These bacteria are resistant to both endogenously produced antibiotics and that encountered in the soil environment. Our first study explored the multiple mechanisms of resistance exhibited by Streptomyces coelicolor against ciprofloxacin, a fluoroquinolone drug. Transcriptomic analysis using whole-genome microarrays suggests that in addition to the up-regulation of target genes, the bacteria counters stress due to antibiotics through up-regulation of efflux pumps and high expression of anti-oxidant genes. Further, growth conditions such as the growth media can modulate gene expression and thus increase the minimum inhibitory concentration of the drug by many folds. Similarly, we have shown that efflux plays an important role in resistance to multiple antibiotics in Mycobacterium smegmatis.
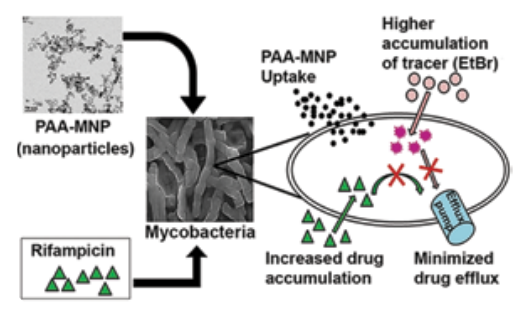
We are also working at ways to counter antibiotic resistance using nano-particles in collaboration with the colloids and nanomaterials group in the department. We have recently demonstrated that a combination of coated iron-oxide (Fe3 O4 ) nanoparticles and anti-TB drugs (for example, rifampicin or isoniazid) can lower the minimum inhibitory concentration required to kill M. smegmatis, and thus overcome intrinsic resistance. While the nanoparticles themselves are not toxic, a synergistic effect is observed, when used in combination with the drug. We have demonstrated, for the first time, through accumulation and efflux kinetics, that nanoparticles inhibit active efflux of molecules from M. smegmatis cells. Further, to mimic clinically resistant strains, we demonstrated that the nanoparticles are effective against laboratoryderived highly drug resistant strains.
Many recombinant therapeutics are complex glycoproteins for which mammalian cells are the preferred expression systems. With the advent of biosimilars, there is a further need to increase the productivity of these cell lines and accelerate process development. We are investigating the role of the protein secretion pathway in limiting productivity of a panel of CHO cell lines that produce monoclonal antibodies at varying productivity levels from 5 pg/cell/day to 50 pg/cell/day. The experiments are coupled with a mathematical model to guide our efforts to modulate the levels of unfolded protein response UPR pathways genes, using siRNA or chemical inhibitors. Further, we wish to understand the effect of process parameters and reactor configurations on cellular stress and productivity